Congratulations | Kossel and Medtronic Create a New Future for Precise Coronary Intervention

January 20, 2026 – The strategic cooperation signing ceremony between Kossel Medtech (Suzhou) Co., Ltd (“Kossel”) and Medtronic (Shanghai) Management Co., Ltd. (“Medtronic”) was successfully held. This collaboration marks the establishment of a deep strategic partnership. Centered on Kossel’s core product, the Seledora® Coronary Scoring Balloon Catheter, the two sides will work together to promote its […]
K-Rhino Peripheral Thrombus Aspiration System
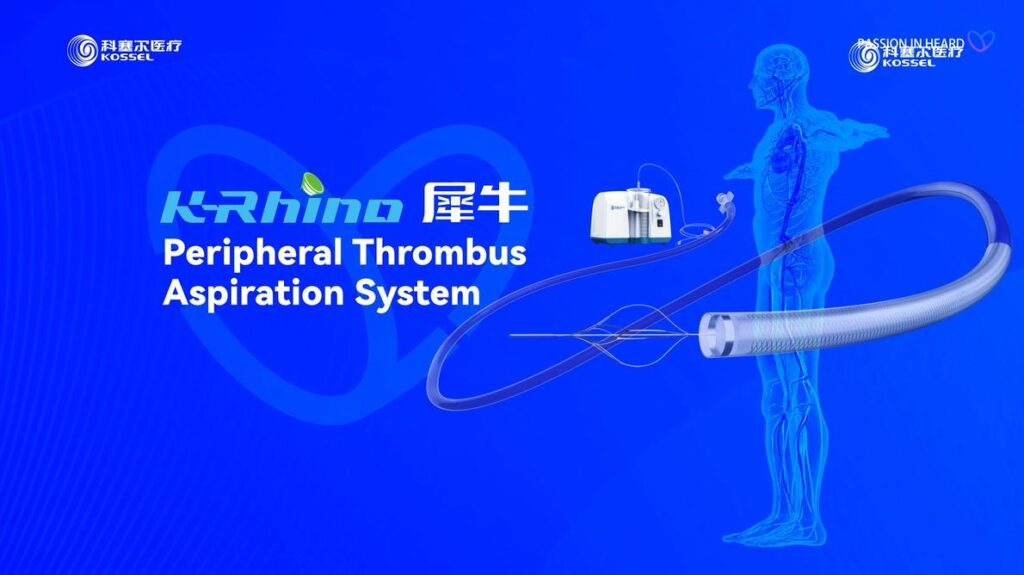
Purpose-built for peripheral thrombus aspiration
Vena Cava Filter Procedure (Series)

How to address the issue that the angle between the inferior vena cava and the left iliac vein is large and there is a risk of tilting during the filter release?
Igniting the Global Medical Stage! Kossel MEDICA 2025 Concludes Successfully!

From November 17th to 20th, MEDICA 2025, the world’s premier medical extravaganza, grandly took place in Düsseldorf, Germany. Amid this feast of ideas gathering global cutting-edge technology and medical wisdom, we outlined the future blueprint of the healthcare industry through frontier technologies, innovative products, and in-depth dialogues. Chinese Solutions on the International Stage Bringing its […]
Vena Cava Filter Procedure (Series)

How to address the issue that the angle between the inferior vena cava and the left iliac vein is large and there is a risk of tilting during the filter release?
Kossel Debuts at CCT 2025, Deepening Penetration in the Asian Market

From October 23 to 24, 2025, the 2025 Complex Cardiovascular Therapeutics (CCT 2025) was grandly held in Kobe, Japan. As one of the most influential academic conferences on cardiovascular interventional therapy in Asia, it shares the latest advancements in PCI therapy, structural heart disease, and surgical treatment through live surgical demonstrations, special seminars, and academic […]
When 8–12F is the mainstream, why do we insist on 14F?
Recently, Kossel Medtech (Suzhou) Co., Ltd. received approval from the National Medical Products Administration (NMPA) for its independently developed Peripheral Thrombectomy Catheter (Registration No. 20253031757). This product, together with the previously approved Medical Electric Suction Pump (Registration No. 20252141564), Disposable Suction Connection Tube (Registration No. 20252141452), Negative Pressure Collection Device (Registration No. 20252109), and the […]
Good News! Asia Market Expansion Accelerates Further! Three PTCA Balloon Catheters Have Obtained Certification from KFDA!

Recently, the three types of PTCA balloon catheters developed independently by Kossel – Selethru II (SC, NC26, Super NC) – have obtained the certificate from the KFDA. Until now, the Selethru PTCA balloon catheter has been approved for sale in Asia, the Middle East, South America, and other regions. With its outstanding clinical performance, it […]
SOLACI SOCIME 2025 | Unlock the Latin American Medical Market and Jointly Build a Global Health Future!

The 2025 Annual Congress of the Latin American Society of Interventional Cardiology (SOLACI SOCIME 2025) was grandly held in Mexico from August 6 to 8. As the most influential cardiovascular intervention event in Latin America, SOLACI brought together experts and scholars in the field of cardiovascular intervention from various countries in Latin America and around […]
Octoparms® Vena Cava Filter Procedure(Series)
Techniques For Femoral Access!