Reference Document | Expert Consensus on Ultrasound Interventional Therapy for Hemodialysis Vascular Access in China (2024 Edition)
Abstract This consensus aims to provide clinical physicians with standardized guidance for ultrasound-guided PTA procedures, offer a reference for healthcare administrators in conducting quality control, and promote the wide application and popularization of ultrasound technology in arteriovenous dialysis access. Preoperative ultrasound assessment Sequence for ultrasound assessment of autologous AVF In the direction of blood flow […]
Seledora® Coronary Scoring Balloon Catheter– Product Overview
“Precise lesion preparation” and “vascular functional restoration” are the main themes in current coronary interventional therapy. Compared with common balloon angioplasty, which compresses plaques in a disordered manner, scoring balloons offer a more targeted solution. On the one hand, the scoring elements focus pressure on the lesion, enabling controlled plaque modification and reducing the risk […]
Seledora Coronary Scoring Balloon Catheter

Seledora® Coronary Scoring Balloon Catheter is Now Available Low-pressure focused dilation, safe and effective, flexible and efficient, helping to fully pre-dilate the coronary artery
Celebratory News丨Selethru® PTCA balloon dilatation catheter has obtained the CE MDR certification

Recently, Kossel’s independently developed Selethru® PTCA balloon dilatation catheter has successfully passed the EU Medical Device Regulation (MDR) certification, marking that the product has met the highest international standards in terms of safety, effectiveness and clinical performance. Europe is a globally important medical device market. The acquisition of MDR certification has provided strong support for […]
Deepening Latin American Ties — Kossel Medtech Strikes a New Chord in the Land of Samba

Hospitalar 2025 | São Paulo, Brazil | May 20–23 Hospitalar, one of South America’s most influential healthcare exhibitions, concluded successfully in São Paulo. Leveraging Brazil’s strategic position in Latin America, the event has become a key platform for regional medical integration and international collaboration. At this major event, Kossel Medtech showcased a portfolio of flagship products […]
Cathios® CARDIAC ABLATION CATHETER

Cathios® CARDIAC ABLATION CATHETER A. Easy Control 1:1 twisting force feedback makes tip placement convenient and quick B. Excellent Tip and Special design Curve Part 16X2 strands wire braid curve part and brilliant tip help catheter stay close to cardiac structure C. Adjust Precisely It is easy to locate the target points of heart with […]
Falspeed® PTA Balloon Dilatation Catheter

PTA balloon catheter is mainly used for limb blood vessels, extended to the arteries, such as renal artery, coronary artery, and vein, such as the expansion of the vena cava stenosis, and treatment of artificial blood vessels, transplant vascular stenosis or occlusion. 1. A wide range of diameters and lengths are available, covering different clinical […]
Cathlink Electrode Lead Introducer Sheath
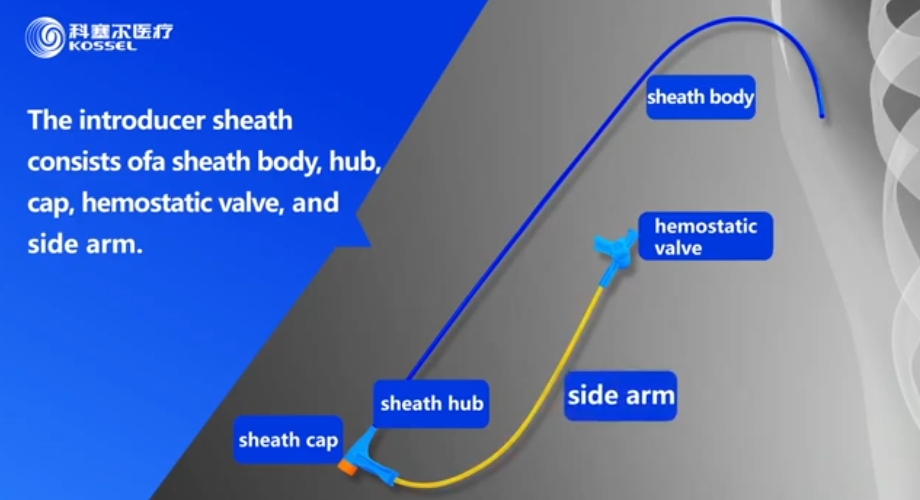
Dedicated sheath for electrode leads – reduces procedural complexity and increases success rates. For more info,please contact us at : Web:https://www.kossel-medical.com/ Email:nicole.ma@kosselmed.com WhatsApp:+86 131 4777 1518
Misatin® Hydrophilic Guidewire

Misatin® Hydrophilic Guidewire Long tapered tip for easy navigation. NiTi mandrel for stability and kink resistance. Tungsten polymer jacket for visibility and control. Hydrophilic coating for smooth passage and reduced injury.
The Clinical Research of Mistent® Coronary Drug Eluting Stent System
一、Stent Design The biodegradable coating (PLGA polymer combined with sirolimus crystals) was realized based on supercritical fluid technology, which gave birth to the Mistent® coronary drug-eluting stent. It has two distinctive features: “Quick Healing” and “Long-lasting Drug Efficacy”. The biodegradable polymer enables rapid endothelialization and reduces chronic inflammation and the occurrence of stent thrombosis; the […]